Stringy stonecrop (Sedum sarmentosum Bunge) of the family Crassulaceae is a mat-forming succulent with creeping and ascending stems. The plant's native range is China, Japan, Korea, Manchuria, Thailand, and Vietnam, and the plant was introduced to America and Europe [1]. In Korea, the plant is grown as a wild vegetable. During disease surveys in spring 2019 and 2020, Sclerotinia rot symptoms were observed in stringy stonecrop plants growing in vinyl greenhouses in Yeoju and Icheon, Gyeonggi Province, Korea. The initial symptoms appeared as soft rot on stems and leaves at or above the soil line. Furthermore, the symptoms progressed upwards, and the infected plant parts exhibited white to grayish-yellow discoloration (Fig. 1A). Abundant cottony mycelia developed on the infected plant parts, and globose to irregular black sclerotia were produced on the infected plant parts at the late stages of the disease development (Fig. 1B). To investigate disease incidence, three sites in a vinyl greenhouse were observed, and 100 plants from each site were examined for disease incidence. As a result, the incidence of diseased plants in the vinyl greenhouses in the two locations was 1‒5% (Table 1).
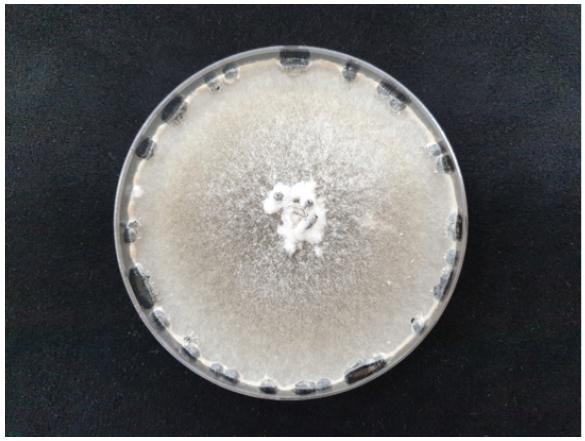
The diseased plants of stringy stonecrop were collected from the vinyl greenhouses, and fungi were isolated from them. The 3–5 mm-long lesion pieces were cut from the diseased stems, surface-sterilized using 1% sodium hypochlorite solution for 1 min, and plated on 2% water agar (WA; FUJIFILM Wako Chemicals, Chuo-Ku, Osaka, Japan). The fungal mycelia growing from the lesion pieces were transferred to potato dextrose agar (PDA; Difco, Sparks, MD, USA) slants after incubating the plates at 22℃ for 1 day. Eight isolates of Sclerotinia sp. were obtained from the lesion pieces. The isolates were cultured on PDA in 9 cm-diameter Petri dishes at 22℃ in the dark for 15 days, and the colony morphology of the isolates grown on the medium was examined. Colonies of the isolates on PDA consisted of white to gray mycelia and globose to irregular black sclerotia (Fig. 2). The number of sclerotia produced in a 15-day-old PDA culture ranged from 26‒33, and the size of sclerotia was 2.0‒12.0 mm×2.0‒4.4 mm. The morphological characteristics of the isolates were similar to those of S. sclerotiorum (Lib.) de Bary described in previous studies [2, 3].
In order to identify the accurate species of all isolates from stringy stonecrop, DNA sequencing of the internal transcribed spacer (ITS) region of rDNA was carried out using the ITS1/ITS4 primer pair [4]. Nucleotide sequences of a total size of 512−524 bp were obtained for each isolate, and the sequences of the eight isolates (SLSC-2035 to SLSC-2042) were deposited in the NCBI GenBank with accession numbers of ON506019‒ON506026. For phylogenetic analysis of the isolates, the neighbor-joining tree was generated with related species of Sclerotinia using MEGA X software [5] and Clarireedia homoeocarpa epitype strain was used as an outgroup. As a result, the ITS region sequences of the eight isolates were 100% identical to those of reference strains of S. sclerotiorum DAOM:241671 and CBS:499.50 in GenBank (Fig. 3). Therefore, all isolates from stringy stonecrop were identified as S. sclerotiorum based on the morphological and molecular characteristics.
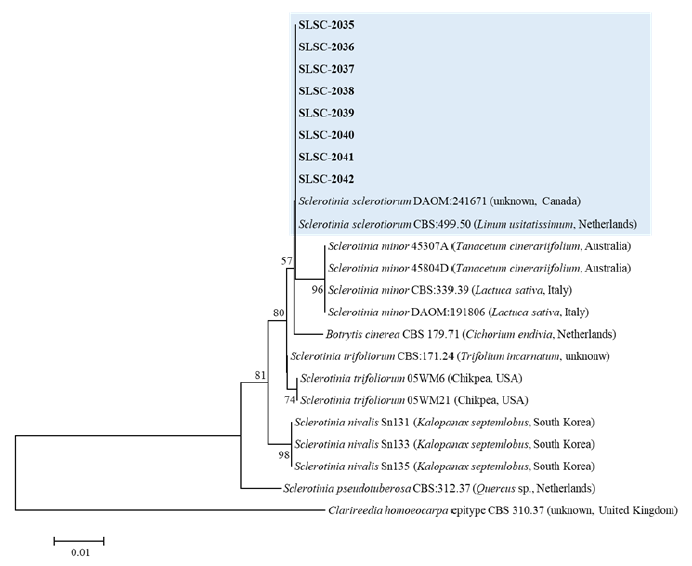
Fig. 3.Phylogenetic tree based on internal transcribed spacer (ITS) region of rDNA for Sclerotinia sclerotiorum isolates from stringy stonecrop and related species. Sequence data were obtained from the NCBI GenBank database. The tree was generated using neighbor-joining analysis and Kimura 2-parameter model. The bar represents the number of nucleotide substitutions per site.
Three isolates of S. sclerotiorum from stringy stonecrop were used to confirm their pathogenicity to the host plant using artificial inoculation. Mycelial disks of 6 mm in diameter cut from the margins of actively growing cultures of each isolate on PDA were placed on the stems at the soil surface level of 45-day-old stringy stonecrop plants grown in circular plastic pots (height: 15 cm; upper diameter: 17 cm; lower diameter: 10 cm) in a vinyl greenhouse. Inoculated plant pots were placed in plastic boxes (60 cm×43 cm×33 cm) under 100% relative humidity at room temperature (22‒24°C) for 4 days. Thereafter, the inoculated plant pots were taken out of the plastic boxes and kept indoors at room temperature (22‒24°C). The pathogenicity of the isolates was rated based on the degree of Sclerotinia rot symptoms 7 days after inoculation. The inoculation test was conducted in triplicate.
All tested isolates of S. sclerotiorum induced Sclerotinia rot symptoms in the inoculated plants (Fig. 1C) but no symptoms were observed in the control plants (Fig. 1D). The symptoms induced by the artificial inoculation of plants were similar to those observed in plants from the vinyl greenhouses, and the inoculated isolates were re-isolated from the lesions.
S. sclerotiorum causes Sclerotinia rot, including stem rot, leaf rot, and fruit rot in many crops [6, 7, 8, 9]. Moreover, white mold disease of stringy stonecrop caused by S. nivalis I. Saito has been reported in China [10]. However, there has been no report of Sclerotinia rot caused by S. sclerotiorum in stringy stonecrop. Therefore, this study is the first report of S. sclerotiorum causing Sclerotinia rot in stringy stonecrop.