1Department of Plant Medicine, Jeonbuk National University, Jeonju 54896, Korea
2Research Center for Plant Medicine, Jeonbuk National University, Jeonju 54896, Korea
3Department of Forestry, Environment, and Systems, Kookmin University, Seoul 02707, Korea
4Division of Environmental Science and Ecological Engineering, Korea University, Seoul 02841, Korea
*Correspondence to iychoi@jbnu.ac.kr; jhpark10@kookmin.ac.kr
Korean Journal of Mycology (Kor J Mycol) 2025 June, Volume 53, Issue 2, pages 57-63.
https://doi.org/10.4489/kjm.2025.53.2.1
Received on March 18, 2025, Revised on May 08, 2025, Accepted on May 09, 2025, Published on June 30, 2025.
Copyright © The Korean Society of Mycology.
This is an Open Access article which is freely available under the Creative Commons Attribution-NonCommercial 4.0 International License (CC BY-NC) (https://creativecommons.org/licenses/by-nc/4.0/).
Two Erysiphe species, E. diervillae and E. weigelae, were recognized powdery mildew pathogens infecting Weigela species in East Asia, with E. weigelae first documented in Korea in the early 2000s. Based on a recent taxonomic revision, E. weigelae was synonymyzed with E. diervillae var. weigelae. To update our knowledge on this fungus, the holomorph morphology of the Korean powdery mildew on Weigela was described by integrating detailed morphological characterization and molecular phylogenetic studies. The newly generated sequences of the internal transcribed spacer regions and large subunit gene of rDNA have been submitted to GenBank. This study provides the first molecular phylogeny report of E. diervillae.
Caprifoliaceae, Erysiphaceae, Erysiphe weigelae, molecular phylogeny, Weigela
Two Erysiphe species, E. diervillae Miyabe ex U. Braun and E. weigelae Z.X. Chen & S.B. Luo, were listed in Braun’s monograph in 1987 as powdery mildew pathogens infecting Weigela spp. [1]. E. diervillae was first introduced by Salmon in 1900, based on the herbarium material collected by Miyabe from Diervilla japonica DC. (currently classified as Weigela hortensis (Siebold. & Zucc.) K. Koch.) in Japan [2]. Jaczewski later reclassified it as a forma of Erysiphe communis (Wallr.) Schltdl. (referred to as E. communis f. diervillae) [3]. Later in 1983, Braun raised it to the species level as E. diervillae, in accordance with its earliest classification [3]. This fungus has long been considered endemic to Japan. Another powdery mildew species, E. weigelae, was introduced in 1984, having been found on W. japonica var. sinica (Rehder) L.H. Bailey, in Fujian, China [4]. In 1990, Heluta reduced it to a variety of E. diervillae, as E. diervillae var. weigelae (Z.X. Chen & S.B. Luo) V.P. Heluta [5]. Based on the current concept, the three varieties of E. diervillae – var. diervillae, var. weigelae, and var. chasanensis Heluta – were classified as powdery mildew pathogens of Weigela spp. [6]. According to Braun and Cook [6], var. weigelae differs from var. diervillae by having smaller chasmothecia (85–130(–140) µm vs. 110–160 µm) and ascospores (15–20 × 8–12 µm vs. 15–36 × 9–17 µm). E. diervillae var. chasanensis, which is known only from W. praecox L.H. Bailey (currently classified as W. florida (Bunge) A.DC.) in Russia, differs from the other two varieties by having irregularly branched ascomatal appendages [6].
In Korea, Erysiphe powdery mildew on W. florida and W. subsessilis L.H. Bailey was initially recorded as E. weigelae in 2003, based on its similarities to the original description of teleomorphic traits [4], and the differences in anamorphic features from those of E. diervillae as described by Nomura [7]. Additionally, the authors reported the presence of secondary hyphae in this fungus for the first time [8].
During our extensive field forays to study phytopathogenic fungi in Korea, 42 samples of powdery mildew on Weigela spp. were collected from various regions across Korea since 1999 and have been preserved in the Korea University herbarium (KUS). Four recently collected samples on W. subsessilis were examined as part of our study: KUS-F31246 (Oct 9, 2019; Gapyeong), F32902 (Jun 3, 2022; Wanju), F33739 (Jun 8, 2023; Jinan), and F34400 (Oct 31, 2024; Namyangju).
Detailed morphological characteristics of the fungus were examined using fresh samples under an Olympus BX50 microscope (Olympus, Tokyo, Japan). Photomicrographs were captured using a Zeiss AX10 microscope equipped with an AxioCam MRc5 camera (Carl Zeiss, Oberkochen, Germany). Mycelial mats with conidiophores and conidia were amphigenous and caulicolous, persistent, in white patches, and occasionally causing reddish discoloration (Fig. 1A–C). Hyphae were superficial, straight to wavy, branched, and 4–8(–10) µm wide. Hyphal appressoria were multi-lobed or moderately lobed, occurring singly or in pairs, and 3–8 µm wide (Fig. 1D–E). Conidiophores were predominantly single or in groups of up to three on a hyphal cell, emerging from the upper part of the mother cells, 56–84 × 9–12 µm, straight at foot-cells, becoming broader or narrower upwards, with slight constrictions at the branching point, and producing conidia singly, followed by 2 straight shorter cells (Fig. 1F–I). Conidia were variable in shape, mostly doliiform to subcylindrical, 28–42 × 12–17 µm with a length/width ratio of 1.8–2.8, devoid of conspicuous fibrosin bodies, and occasionally guttulate (Fig. 1J); primary conidia were asymmetrical at both ends, exhibiting an apically rounded and basally subtruncate morphology (Fig. 1K and L); germ tubes were produced on the perihilar position of the conidia and variable in shape (Fig. 1M–O). Chasmothecia were gregarious and 110–136 µm in diameter; peridium cells were irregularly polygonal and 8–20 µm in diameter (Fig. 1P and Q); chasmothecial appendages were located in the lower half, mycelioid, simple, straight when young, becoming geniculate and wavy as they matured, interlaced with each other and the surrounding hyphae, with a variable length reaching up to 4 times the chasmothecial diameter, 3–6 µm wide, usually aseptate, occasionally with a septum at the base, and with thin, smooth, and hyaline walls (Fig. 1P and Q). Asci were 8–16 per chasmothecium, ellipsoid-obovoid, saccate, 50–68 × 30–40 µm, shortstalked or subsessile, 8-spored, with a 1–1.5 µm thick wall (Fig. 1R). Ascospores were ellipsoid, 15–18 × 9–10 µm with a length/width ratio of 1.5–2.1, and hyaline to faintly yellow (Fig. 1S). These characteristics were consistent with the description of Erysiphe diervillae, and based on the size of the chasmothecia and ascospores, our isolates were identified as Erysiphe diervillae var. weigelae [6].
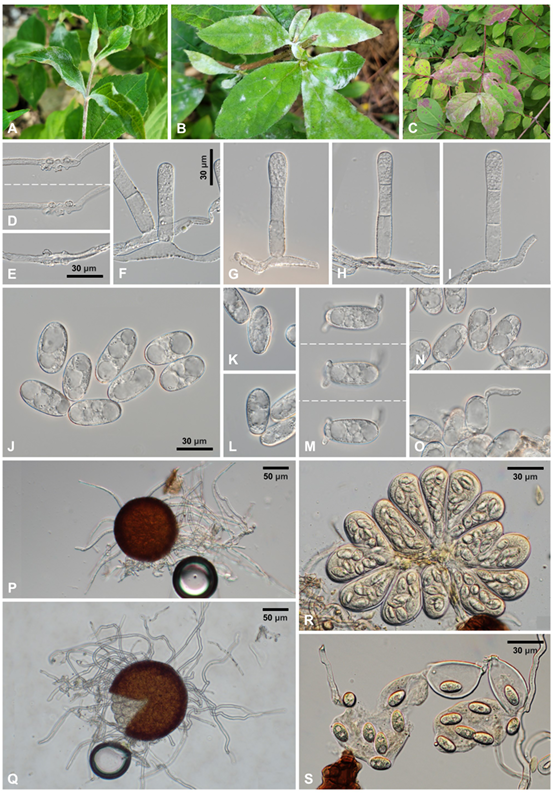
Fig. 1. Erysiphe diervillae on Weigela subsessilis. A–B: Typical powdery mildew symptoms on leaves and herbaceous stems. C: Reddish discoloration of the infected leaf areas. D: Appressoria formed on the hypha, focused at different depths. E: Appressoria formed on the hypha. F–I: Conidiophores. J: Conidia. K–L: Primary conidia. M: Conidium having three germ tubes, focused at different depths. N–O: Conidia in germination. P: Young chasmothecium with short appendages. Q: Mature chasmothecium with long appendages and protruding asci. R: A cluster of asci from a chasmothecium. S: Ascospores.
To investigate the phylogenetic position of the fungus, the nucleotide sequences of the internal transcribed spacer regions (ITS1 and ITS2) including the 5.8S gene, and the large subunit (LSU) gene of the rDNA from the three aforementioned specimens were determined as outlined by Choi et al. [9]. The consensus sequences obtained from assembling the amplicons of the forward and reverse reads were submitted to GenBank (Accession numbers: PV276258, PV276273, and PV276296 for ITS, and PV276297, PV276302, and PV277965 for LSU). To perform phylogenetic analysis, a combined dataset of ITS+LSU was created in MEGA11 and then aligned using the Multiple Sequence Comparison by Log-Expectation (MUSCLE) command [10]. Erysiphe glycines was selected as the outgroup. The maximum parsimony (MP) tree was constructed in PAUP* 4.0a using the heuristic search option with the ʻtree-bisection-reconstruction’ algorithm with 100 random sequence additions to determine the global optimum tree. All sites were considered as unordered and unweighted, and gaps were treated as missing data [11]. Tree scores, such as tree length, consistency index, retention index, and rescaled consistency index, were also calculated. The best tree was selected using the Kishino-Hasegawa and Shimodaira-Hasegawa topology tests in PAUP, and the resulting tree was presented in this study. Maximum likelihood (ML) analysis was implemented in raxmlGUI 2.0.14 using the gamma-distributed rate heterogeneity (GAMMA) model and general time reversible (GTR) substitution [12]. The robustness of the branches was evaluated using bootstrap analyses (BS) through 1,000 replications in both MP and ML analyses.
The ITS and LSU sequences from all three samples were identical. Genetic identity of the sequences was determined by conducting a standard nucleotide basic local alignment search tool (BLASTn) against reference sequences in GenBank. The ITS sequences showed 99.8% identity with E. weigelae (AB015932) and 99.6% to E. diervillae (LC010087). The LSU gene sequences showed approximately 98% similarity to the E. aquilegiae group, including 98.4% to E. caricae-papayae (LC371319), and 98.3% to E. neolycopersici (LC371320) and E. aquilegiae var. ranunculi (AB022405, MT309771). Three sequences for the Erysiphe-Weigela association were available in GenBank; two of them, AB015931 (596 bp) and AB015932 (588 bp), corresponded to the ITS1-5.8S-ITS2 regions and the beginning of the LSU, whereas LC010087 (1,333 bp) contained the complete ITS and a partial 28S rRNA (LSU) gene. The LSU gene of our isolates (978 bp) was manually compared with LC010087, which contains the LSU gene of 775 bp in length. Our sequences were completely identical to LC010087 within the 775 bp nucleotide region. Furthermore, the ITS sequences of AB015931 and LC010087 were identical; however, our sequences showed a two-bp difference at the end of the ITS1 when compared with the other two sequences. The final dataset of ITS+LSU consisted of 44 closely related sequences and 1,503 characters, of which 108 (7.18%) were variable and parsimony-uninformative, and 171 (11.37%) were informative for parsimony analysis. In the MP tree, the three newly obtained sequences clustered in a distinct clade with three E. diervillae sequences, supported by the highest (100/100%) BS values in the MP and ML analyses, respectively (Fig. 2).
Weigela Thunb., a genus in the family Caprifoliaceae, comprises deciduous shrub species native to East Asia and widely cultivated for their ornamental and medicinal value [13]. A phylogenetic analysis of this genus revealed that Weigela belongs to the Diervilloideae group, which occupies the basal position within Caprifoliaceae, suggesting its early divergence from other genera in the family [13]. Therefore, relevant sequences were included in our analyses to assess the influence of plant evolution on the phylogenetic relationships between Weigela powdery mildew and other powdery mildews infecting caprifoliaceous hosts (Abelia, Lonicera, and Symphoricarpos). These results strongly supported the phylogenetic placement of Weigela, consistent with E. diervillae, distinguishing E. diervillae from other Erysiphe powdery mildews on caprifoliaceous plants (Fig. 2).
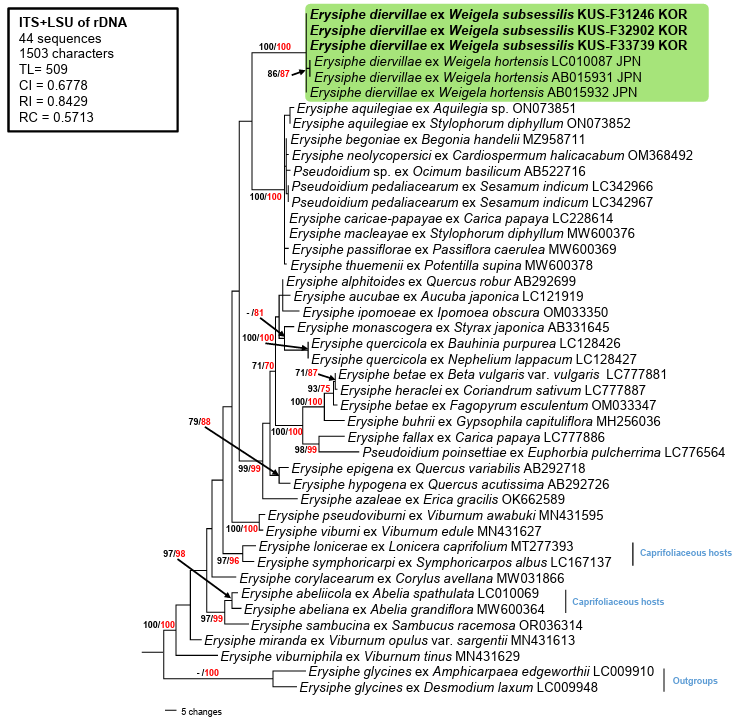
Fig. 2. A maximum parsimony tree for Erysiphe diervillae generated using a combined dataset of ITS+LSU sequences of 44 strains. Two strains of Erysiphe glycines were selected as outgroups. The isolates obtained in this study are indicated in bold. Bootstrap values (≥70%) obtained in the maximum parsimony (black) and maximum likelihood (red) analyses were indicated on the related branches. The calculated tree scores were given in the box. ITS, internal transcribed spacer; LSU, large subunit; TL, tree length; CI, consistency index; RI, retention index; RC, rescaled consistency index.
Five species and one variety of Weigela (W. coraeensis Thunb., W. decora (Nakai) Nakai, W. decora var. amagiensis (Nakai) H. Hara, W. florida, W. hortensis, and W. subsessilis) are distributed in Korea [https://species.nibr.go.kr]. Among them, E. diervillae has been reported on W. f lorida and W. subsessilis. The distribution of E. diervillae is confined to Japan, China, Korea, and Far East Russia so far [14]. Since the introduction of Weigela spp. in Europe, more than 200 cultivars have been developed for ornamental purposes over the past two centuries [15]. Given the widespread use of Weigela species, cultivars, and hybrids in landscaping worldwide, the detailed information on E. diervillae would considerably contribute to effective disease monitoring efforts and aid in the development of disease-resistant cultivars.
The authors declare no competing interests.
This study was supported by the R&D Program for Resolving Current Issues in Forest Disaster and Damage (Project No. RS-2024-00403034), funded by the Korea Forest Service (Korea Forestry Promotion Institute).
1. Braun U. A monograph of the Erysiphales (powdery mildews). Nova Hedwigia 1987;89:1700.
2. Salmon ES. A monograph of the Erysiphaceae. Memoirs of the Torrey Botanical Club 1900;9:1-292. [DOI]
3. Braun U. Descriptions of new species and combinations in Microsphaera and Erysiphe (IV). Mycotaxon 1983;18:113-29. [DOI]
4. Chen ZX, Gao RX, Luo SB, Liu BC. New species of powdery mildew from Wuyishan. Acta Mycol Sin 1984;3(2):75-80.
5. Heluta VP. Novi dlya ta CRCR taksoni boroshnosto-rosyanikh gribiv iz zapovidnika “Kedrova Pad” (Primorsk’ky Kray). Ukrayins’kyi Botanichnyi Zhurnal 1990;47(5):79-83.
6. Braun U, Cook RTA. Taxonomic manual of the Erysiphales (powdery mildews). CBS Biodiversity Series 11. Utrecht: Centraalbureau voor Schimmelcultures; 2012.
7. Nomura Y. Taxonomic study of Erysiphaceae of Japan. Tokyo: Yokendo Ltd.; 1997.
8. Shin HD, Choi YJ. First record of Erysiphe weigelae from Korea and its anamorph. Mycotaxon 2003;86:269-75. [DOI]
9. Choi IY, Abasova L, Choi JH, Park JH, Shin HD. Erysiphe cornicola, a powdery mildew occurring on Cornus controversa in Korea. Kor J Mycol 2023;51:57-62. [DOI]
10. Tamura K, Stecher G, Kumar S. MEGA11: molecular evolutionary genetics analysis version 11. Mol Biol Evol 2021;38:3022-7. [DOI]
11. Swofford DL. PAUP: phylogenetic analyses using parsimony (and other methods). Version 4.0b10. Sunderland: Sinauer Associates; 2002.
12. Silvestro D, Michalak I. raxmlGUI: a graphical front-end for RAxML. Org Divers Evol 2012;12:335-7. [DOI]
13. Wang L, Li F, Zhao K, Yang J, Sun H, Cui X, Dong W, Li E, Wang N. Comparative plastomes sheds light on phylogeny of Weigela. Front Plant Sci 2024;15:1487725. [DOI]
14. Farr DF, Rossman AY. Fungal databases, US National Fungus Collections [Internet]. Maryland: Agricultural Research Service, US Department of Agriculture; 2025 [cited 2025 Mar 7]. Available from https://fungi.ars.usda.gov/.
15. Hobloss S, Bruguière A, Champy-Tixier AS, Miyamoto T, Tanaka C, Dessertaine S, Sautour M, Lacaille-Dubois MA, Mitaine-Offer AC. Oleanane-type glycosides from Weigela x Styriaca and two cultivars of W. florida: “Minor Black” and “Brigela.” Phytochem Lett 2022;50:77-84. [DOI]