1Department of Mushroom Science, Korea National University of Agriculture and Fisheries, Jeonju 54874, Korea
2Mush&, Jeonju 54874, Korea
3farmOLIN, Pyeongtaek 17716, Korea
*Correspondence to mshy1219@af.ac.kr
Korean Journal of Mycology (Kor J Mycol) 2025 June, Volume 53, Issue 2, pages 121-125.
https://doi.org/10.4489/kjm.2025.53.2.7
Received on May 30, 2025, Revised on June 11, 2025, Accepted on June 11, 2025, Published on June 30, 2025.
Copyright © The Korean Society of Mycology.
This is an Open Access article which is freely available under the Creative Commons Attribution-NonCommercial 4.0 International License (CC BY-NC) (https://creativecommons.org/licenses/by-nc/4.0/).
Cordyceps militaris is widely cultivated in East Asia as an edible and medicinal mushroom. Although fungal infections affecting C. militaris have been reported in several countries, no such outbreaks have been documented in South Korea. In the present study, we investigated an occurrence of fungal infection of C. militaris at a farm in Pyeongtaek, South Korea. Infected fruiting bodies were initially covered with white cottony mycelia that later developed into yellowish-green colonies. Two fungal strains were isolated from the infected fruiting bodies and identified as Chlorocillium lepidopterorum based on morphological characteristics and phylogenetic analysis using a multigene dataset (the large subunit of nuclear ribosomal RNA gene, internal transcribed spacer regions, translation elongation factor 1-alpha, and RNA polymerase II second largest subunit). Pathogenicity of the strains was confirmed by artificial inoculation. The symptoms of the strains were consistent with those observed in naturally infected fruiting bodies on farms. This is the first report of C. militaris infection caused by C. lepidopterorum in South Korea.
Chlorocillium lepidopterorum, Cordyceps militaris, Mushroom diseases, Unrecorded species
Cordyceps militaris (L.) Fr., belonging to the order Hypocreales, is well known as an important edible and medicinal mushroom in China, Korea, and Japan [1]. Cordyceps militaris has been used as a health supplement and crude drug in East Asia due to various bioactive compounds such as cordycepin, polysaccharides, and ergosterol [2,3]. Cordyceps militaris has traditionally been harvested from the wild, making it extremely rare and difficult to obtain. However, it is currently cultivated in China, Japan, and Korea [3].
Fungal diseases have been reported during commercial production of C. militaris. The primary causative agents are, to date, Calcarisporium cordycipiticola in China [4] and Lecanicillium coprophilum in Vietnam [5]. Although C. militaris is continuously cultivated in Korea, no serious fungal disease outbreaks have been reported. However, recently, fungi that form white cottony colonies have been frequently observed on the fruiting bodies of C. militaris, leading to substantial losses in both quality and yield. Accordingly, we investigated and characterized the infectious mycoparasite affecting C. militaris in South Korea.
Samples were collected from the infected fruiting bodies of C. militaris grown on sterilized oats at the farmOLIN, Pyeongtaek, on March 16, 2023, and April 23, 2023. The fruiting bodies initially displayed white mycelia, which gradually developed into yellowish-green cottony mycelial colonies (Figs. 1A and B). Infected fruiting bodies were transferred to potato dextrose agar (PDA; Difco, Becton Dickinson) plate and incubated at 25℃ for 7 days. Fungal colonies grown from the inoculated samples were transferred to fresh PDA plates. The two isolated strains were preserved in a metabolically inactive state (20% glycerol at –80°C) at the Korea National University of Agriculture and Fisheries.
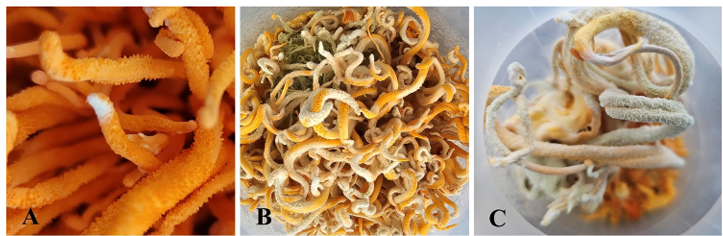
Fig. 1. Fungal pathogens on fruiting bodies of Cordyceps militaris. (A) Initial formation of white mycelium on the fruiting bodies. (B) Progression to yellowish-green mycelial colonies on the fruiting bodies. (C) Symptoms of artificial inoculation with C. lepidopterorum MF340.
Genomic DNA was extracted from the isolated strains using an AccuPrep Genomic DNA Extraction Kit (Bioneer, Daejeon, Korea). The PCR amplification was performed using the following primers: LROR and LR5 [6] for the large subunit of nuclear ribosomal RNA gene (LSU), ITS1F [7], and ITS4 [8] for the internal transcribed spacer (ITS) regions, 983F and 2218R [9] for translation elongation factor 1-alpha (tef1α), and fRPB2-5F and fRPB2-7cR [10] for the RNA polymerase II second largest subunit (RPB2). The amplified PCR products were sequenced using the corresponding PCR primers at Bioneer (Daejeon, Korea). The obtained sequences were manually curated with MEGA5 [11] and submitted to GenBank (accession numbers: PV110822-PV110823 for LSU, PV110820-PV110821 for ITS, PV134892– PV134893 for tef1α, and PV134894–PV134895 for RPB2). Multiple sequence alignments were performed using MAFFT v7 [12] with default parameters. Sequence similarity analysis for each strain was conducted across the three loci using MEGA5 [11]. A maximum likelihood phylogenetic tree was generated using RAxML [13] through the CIPRES web portal [14], employing the GTR + GAMMA substitution model with 1,000 bootstrap replicates to evaluate branch support. On the basis of the concatenated datasets (LSU, ITS, tef1α and RPB2), MF340 (NIBRFGC000510859) and MF354 grouped with C. lepidopterorum SD05361 (type strain) and C. lepidopterorum SD05362 (bootstrap support = 93%; sequence similarity for LSU = 100%, ITS = 100%, and tef-1α = 99.9–100%) (Fig. 1). Pseudometarhizium has been reported as a new genus within Clavicipitaceae, two species have been identified-Pseudometarhizium araneogenum and Pseudometarhizium lepidopterorum-based on morphological, phylogenetic, and ecological characteristics.
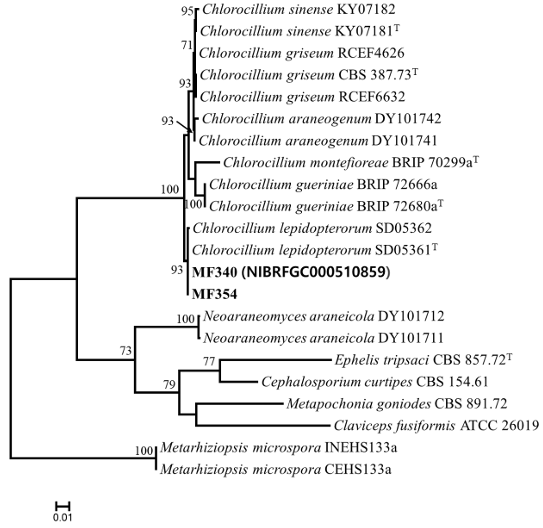
Fig. 2. Maximum likelihood phylogenetic tree based on the concatenated datasets (LSU, ITS, tef-1α, and RPB2) used to identify Chlorocillium strains from infected fruiting bodies of Cordyceps militaris. Bootstrap scores of >70 are presented at the nodes. The scale bar indicates the number of nucleotide substitutions per site. “T” indicates the ex-type strains. Strains reported in the current study are represented in bold.
P. araneogenum and P. lepidopterorum have been isolated from spiders and insects, respectively [15]. Recent phylogenetic analyses using multigene datasets (LSU, ITS, tef-1α, and RPB2) have indicated that the genus Pseudometarhizium is synonymous with Chlorocillium; thus, the two previously recognized species have been reclassified under Chlorocillium [16]. We follow this nomenclature in the present study and report, for the first time, Chlorocillium lepidopterorum infection of C. militaris.
In the pathogenicity test of C. lepidopterorum MF340, three healthy fruiting bodies of C. militaris from the farmOLIN were sprayed with 5 mL of conidial suspensions (1 × 105 conidia/mL) and then incubated at 25℃. Initially, white mycelia were observed on the fruiting bodies, and these mycelia eventually formed yellow-green mycelial colonies throughout the fruiting bodies (Fig. 1C). The fungus in the inoculated cultures was reisolated and identified as C. lepidopterorum based on sequence analysis.
The colony and microscopic features of C. lepidopterorum was observed on PDA after incubation for 14 d at 25℃. Color names and alphanumeric codes followed the Methuen Handbook of Color [17]. The microscopic features were examined under a light microscope (DM2000, Leica, Germany).
Description: Colony diam, 14 d, in mm: PDA 20℃ 15-16; PDA 25℃ 16-18; PDA 30℃ no growth. Colony characteristics: white to orange-white (5A2), velvety to floccose, and reverse color greyish orange (5B3). Hyphae hyaline, smooth, and septate (Fig. 3A, B). Conidiophores typically emerge from aerial hyphae. Phialides occur singly or in two to three groups, with a cylindrical basal that gradually narrows into a short, well-defined neck, 17.5–30.3 × 1.2–1.5 μm. Conidia, hyaline, fusiform, smooth-walled, 3.1–4.5 × 1.4–1.6 μm (Fig. 3C–E).
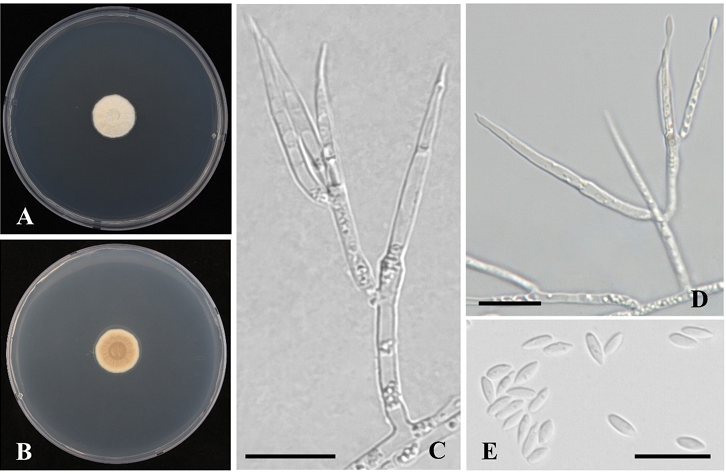
Fig. 3. Chlorocillium lepidopterorum MF340. A-B: Obverse (A) and reverse (B) of colony on potato dextrose agar (PDA) after incubation for 14 d at 25℃. C, D: Conidiophores; E: Conidia (scale bar: C–E = 10 μm).
Strains examined: Jinwi-myeon, Pyeongtaek-si, Gyeonggi-do, Republic of Korea, isolated from the fruiting bodies of C. militaris; strains MF340 (NIBRFGC000510859) and MF354.
Note: Chlorocillium lepidopterorum is phylogenetically close to C. araneo genum, C. griseum and C. lepidopterorum within Clavicipitaceae. Chlorocillium lepidopterorum is easily distinguished from other species within Clavicipitaceae by size of phialide and conidia [16]. Chlorocillium lepidopterorum can be distinguished from C. araneo genum (8.3–23.3 × 1.3–2.2 μm) by slight larger phialides. Chlorocillium lepidopterorum produces smaller conidia than C. griseum (4.5–6 × 1.0–1.5 μm).
No conflicts of interest were reported by the authors.
This research was supported by grants from the National Institute of Biological Resources (NIBR) funded by the Ministry of Environment (MOE) of the Republic of Korea (NIBR202002104 and NIBR202511101).
1. Olatunji OJ, Tang J, Tola A, Auberon F, Oluwaniyi, O, Ouyang Z. The genus Cordyceps: An extensive review of its traditional uses, phytochemistry and pharmacology. Fitoterapia 2018;129:293-316. [DOI]
2. Shweta, Abdullah S, Komal, Kumar A. A brief review on the medicinal uses of Cordyceps militaris. Pharmacol Res Mod Chin Med 2023;7:100228. [DOI]
3. Das SK, Masuda M, Sakurai A, Sakakibara M. Medicinal uses of the mushroom Cordyceps militaris: Current state and prospects. Fitoterapia 2010;81:961-8. [DOI]
4. Sun JZ, Dong CH, Liu XZ, Liu JK, Hyde KD. Calcarisporium cordycipiticola sp. nov., an important fungal pathogen of Cordyceps militaris. Phytotaxa 2016;268:135-44. [DOI]
5. Nguyen TT, Le TNG, Nguyen TH. First report of emerging fungal pathogens of Cordyceps militaris in Vietnam. Sci Rep 2023;13:17669. [DOI]
6. Vilgalys R, Hester M. Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. J Bacteriol 1990;172:4238-46. [DOI]
7. Gardes M, Bruns TD. ITS primers with enhanced specificity for basidiomycetes-application to the identification of mycorrhizae and rusts. Mol Ecol 1993;2:113-8. [DOI]
8. White TJ, Bruns T, Lee S, Taylor J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis IMA, Gelfand DH, Sninsky JJ, White TJ, editors. PCR protocols: A guide to methods and applications. New York: Academic Press; 1990. p. 315-22. [DOI]
9. Rehner SA, Buckley E. A Beauveria phylogeny inferred from nuclear ITS and EF1-α sequences: Evidence for cryptic diversification and links to Cordyceps teleomorphs. Mycologia 2005;97:8498. [DOI]
10. Liu YJ, Whelen S, Hall BD. Phylogenetic relationships among Ascomycetes: Evidence from an RNA polymerase II subunit. Mol Biol Evol 1999;16:1799-808. [DOI]
11. Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S. MEGA5: Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 2011;28:2731-9. [DOI]
12. Katoh K, Standley DM. MAFFT multiple sequence alignment software version 7: Improvements in performance and usability. Mol Biol Evol 2013;30:772-80. [DOI]
13. Stamatakis A. RAxML-VI-HPC: Maximum likelihood-based phylogenetic analyses with thousands of taxa and mixed models. Bioinformatics 2006;22:2688-90. [DOI]
14. Miller MA, Pfeiffer W, Schwartz T. Creating the CIPRES science gateway for inference of large phylogenetic trees. 2010 Gateway Computing Environments Workshop (GCE); 2010 Nov 13-19; New Orleans, LA, USA. Washington DC: IEEE Computer Society; 2010. p. 1-8. [DOI]
15. Chen WH, Liang JD, Ren XX, Zhao JH, Han YF, Liang ZQ. Phylogenetic, ecological and morphological characteristics reveal two new spider-associated genera in Clavicipitaceae. MycoKeys 2022;91:49-66. [DOI]
16. Chen WH, Li D, Liang JD, Ren XX, Zhao JH, Han YF. Chlorocillium sinense sp. nov. (Clavicipitaceae) and Calcarisporium guizhouense sp. nov. (Calcarisporiaceae) in Hypocreales from China. MycoKeys 2024;109:91-107. [DOI]
17. Kornerup A, Wanscher JH. Methuen handbook of colour. 3rd ed. London: Methuen Publishing; 1978.