Yu-Ra Bae1, Ju-Kyeong Eo2, and and Ahn-Heum Eom1*
1Department of Biology Education, Korea National University of Education, Cheongju, 28173, Korea
2National Ecosystem Survey Team, Division of Ecological Survey, National Institute of Ecology, Seocheon-gun, 33657, Korea
*Correspondence to eomah@knue.ac.kr
Korean Journal of Mycology (Kor J Mycol) 2025 December, Volume 53, Issue 4, pages 237-243.
https://doi.org/10.4489/kjm.2025.53.4.1
Received on October 17, 2025, Revised on November 10, 2025, Accepted on November 10, 2025, Published on December 31, 2025.
Copyright © The Korean Society of Mycology.
This is an Open Access article which is freely available under the Creative Commons Attribution-Non-Commercial 4.0 International License (CC BY-NC) (https://creativecommons.org/licenses/by-nc/4.0/).
Thinning is widely used to regulate tree growth and stand structure. However, its impact on belowground ectomycorrhizal (ECM) fungi in Korean pine forests remains insufficiently documented. Therefore, in this study, we investigated the effects of thinning intensity on ECM fungal community composition and diversity in Pinus densiflora plantations. The ECM root tips were identified using morphological criteria and DNA sequencing. Across intensities, 23–35 ECM fungal species were recorded per plot, and only three taxa, Amanita pantherina, Lactarius sp., and Russula mairei, occurred consistently across all treatments. Non-metric multidimensional scaling indicated a distinct community composition in the control group relative to those in the thinned plots; communities partially overlapped but shifted along the thinning-intensity gradient. These results suggest that thinning intensity influences the ECM fungal community and should be considered when planning forest management that accounts for belowground biodiversity.
Community structure, Ectomycorrhizal fungi, Pinus densiflora, Thinning
Ectomycorrhizal (ECM) fungi play an essential role in forest ecosystems by forming mutualistic associations with plant roots [1]. They produce a fungal mantle that covers the root surface and extend their hyphae into the soil, improving the host’s uptake of nutrients, such as nitrogen and phosphorus. Within the root cortex, fungal hyphae develop a network-like structure known as the “Hartig net,” which mediates the exchange of nutrients between the host and fungus [2]. Through this relationship, host plants transfer a substantial portion of their photosynthetic carbon (~22% of net primary production) to their fungal partner [3]. In return, ECM fungi enhance host resistance to drought, soil acidification, and root pathogens, thereby contributing to plant growth and forest stability [4,5].
Pinus densiflora Siebold et Zucc. is a major ECM tree species that is widely distributed across East Asia, including Korea, Japan, and China. In Korea, P. densiflora plantations play a central role in reforestation, timber production, and the cultivation of economically valuable non-timber products such as Tricholoma matsutake (S.Ito & Imai) Singer [6]. However, the dominance of monospecific pine stands has been associated with a decline in biodiversity and the weakening of ecosystem functions [7]. In managed pine stands, thinning is often performed to mitigate the negative effects of high stand densities. Thinning can reduce density-driven competition and improve understory diversity, light availability, and soil nutrient cycling, thereby enhancing ecosystem functions. These ecological benefits justify using thinning as a management tool to improve biodiversity and maintain forest health.
Various ECM fungi, including Russula, Lactarius, Cortinarius, and Suillus, have been reported in P. densiflora forests in Korea [8]. The composition and diversity of ECM fungal communities vary depending on host specificity and environmental factors, such as soil chemistry, microclimate, and forest management. Among these factors, forest disturbances strongly influence ECM fungal community dynamics. For example, Suillus species commonly dominate the early successional stages after thinning or fire, whereas Russula and Cortinarius species, which are adapted to mature forest conditions, tend to prevail in later stages [9].
Thinning is a key silvicultural practice that is applied to reduce stand density and promote the growth of healthy trees. It improves light penetration, soil moisture, and nutrient redistribution, enhancing forest productivity and stability [10,11]. However, thinning is also a form of anthropogenic disturbance that may affect belowground biotic communities, including that of ECM fungi, which are crucial for forest ecosystem functioning. Despite its ecological importance, the influence of thinning intensity on ECM fungal communities in Korean P. densiflora plantations remains poorly understood. Therefore, in this study, we investigated the effects of thinning intensity on the composition and diversity of ECM fungal communities in P. densiflora plantations.
Sampling was conducted in a P. densiflora plantation on Mt. Chilbo, Byeonggok-myeon, Yeongdeokgun, Gyeongsangbuk-do, Korea (36°38′N, 129°22′E). The stand (~17.5 ha) was thinned in 2008. Within the same stand, an unthinned control plot and five thinned plots adjacent to the control plot were delineated (Table 1). Three plots underwent low thinning (from below) at different intensities: light (34%), moderate (45%), and heavy (60%). Two plots underwent moderate-intensity thinning using different thinning methods: selection thinning (43%) and mechanical thinning (46%).
Table 1. Introduction of collection sites
테이블
To collect ECM material, soils were sampled in March 2016. In the unthinned control plot and each of the five thinned plots, three 10 m × 10 m quadrats were established and overlaid with a 1 m grid (100 cells). Using simple random sampling, five soil cores (100 mL each) were collected per quadrat using a soil sampler (Heungjin Co., Gimpo, Korea), yielding 90 soil samples in total (6 plots × 3 quadrats × 5 cores).
Fine roots recovered from the soil were rinsed with distilled water, and ECM colonization was confirmed under a dissecting microscope based on the presence of a fungal mantle. The ECM root tips were then assigned to morphotypes based on standard criteria, including mantle color and texture, tip morphology and branching pattern, and the presence or absence of emanating hyphae and rhizomorphs [12].
The ECM morphotypes (root tips) were identified using a molecular approach. Root tips were surfacesterilized with 30% H2 O2 and finely homogenized, and total genomic DNA was extracted using the DNeasy Plant Mini Kit (Qiagen, Hilden, Germany) following the manufacturer’s instructions. The nuclear ribosomal internal transcribed spacer (ITS) region (ITS1–5.8S–ITS2), a standard fungal barcode, was amplified with the fungus-specific primer ITS1F (5′-CTTGGTCATTTAGAGGAAGTAA-3′) and the universal primer ITS4 (5′-TCCTCCGCTTATTGATATGC-3′) [13]. We performed PCR amplification under the following conditions: an initial denaturation at 94 °C for 3 min; 35 cycles of 94 °C for 30 s, 50 °C for 30 s, and 72 °C for 90 s; and a final extension at 72 °C for 5 min. The PCR products were purified and Sanger sequenced by SolGent (Daejeon, Korea). The resulting ITS sequences were queried using BLASTn against the National Center for Biotechnology Information nucleotide database to identify the closest matches, and species-level assignments were further verified through phylogenetic analyses conducted using MEGA software [14] (Table S1).
The composition of the ECM fungal community was examined in the unthinned and thinned plots. The Shannon diversity index [15], evenness [16], and species richness were calculated based on the number of species, and the number of ECM root tips was calculated based on the root-tip counts per species and total root-tip abundance using MVSP (Kovach Computing Services, Pentreath, UK). The relative abundance of each species was also determined. Similarity in ECM fungal community structure was assessed using nonmetric multidimensional scaling (NMDS) with the Sørensen (Bray–Curtis) distance measure in PC-ORD 6 (Wild Blueberry Media, Corvallis, OR, USA). To evaluate the effects of thinning intensity, ECM fungal communities were compared between the unthinned control and low-thinning treatments with different thinning intensities using one-way analysis of variance (ANOVA) in SPSS 12 (SPSS Inc., Chicago, IL, USA). To assess the effects of thinning method, ECM fungal community characteristics were compared between the unthinned control and the moderate-thinning treatment that differed only in thinning method (low thinning, selection thinning, and mechanical thinning).
The ECM fungi from the control and treatment plots were identified through morphotyping and molecular analyses. In the unthinned plot, 23 species were recorded. Among the low-thinning treatments, 22 species occurred under light thinning, 32 species under moderate thinning, and 27 species under heavy thinning (Table S1). At the same moderate-thinning intensity, 35 species were found in the selectionthinning plots and 29 species were found in the mechanical-thinning plots. Only three taxa, Amanita pantherina, Lactarius sp., and Russula mairei, occurred in all plots, whereas overall, the species composition differed among treatments, indicating high turnover.
The dominant taxa varied across plots. For example, Lactarius sp.1 had the highest relative abundance in the moderate-thinning plots (52.26%), whereas Clavulina sp.1 was the most abundant in the heavy-thinning plots (10.96%). Meanwhile, Tomentella spp. and Russula mairei occurred frequently in several plots, with R. mairei reaching its highest abundance under moderate thinning (20.80%). These dominant species differed among the treatments, contributing to high turnover in ECM fungal community structure (Table S1).
We performed NMDS to assess among-plot differences in the ECM fungal community composition (Fig. 1). The ECM fungal communities differed with thinning intensity, and the unthinned control plot was clearly distinct from the thinned plots along Axis 1. Although there was some overlap among treatments, the ordination indicated a gradual shift in community structure along the thinning-intensity gradient. The mechanically thinned plot showed low within-group dispersion and was separated from the other treatments, whereas the selection-thinned plot had more overlap with the control. In contrast, the moderately thinned plots exhibited greater within-group dispersion in the ordination, likely reflecting the increased microhabitat heterogeneity created by partial canopy opening. Such heterogeneity in soil moisture, litter input, and root conditions could lead to more variable ECM assemblages among the plots in this treatment. This broader range of assemblages under moderate thinning is consistent with the higher alpha diversity (Fig. 2), indicating that intermediate levels of disturbance create favorable conditions for diverse ECM fungal communities. Overall, thinning was associated with a directional shift in the ECM fungal community composition, whereas the unthinned stand retained a composition characteristic of undisturbed conditions.
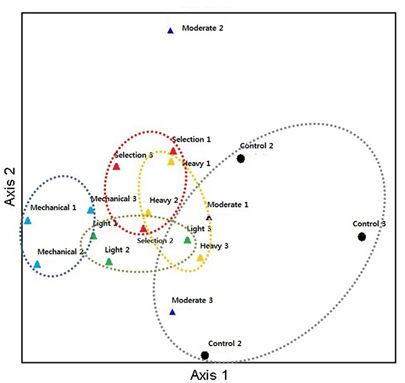
Fig. 1. Non-metric multidimensional scaling (NMDS) ordination of ectomycorrhizal (ECM) fungal communities across control and thinning treatments (Sørensen/Bray–Curtis). Points are plots; dotted envelopes indicate groups. Intensity: green (light), blue (moderate), yellow (heavy). Method at moderate intensity: cyan (mechanical), red (selection). Control = black.
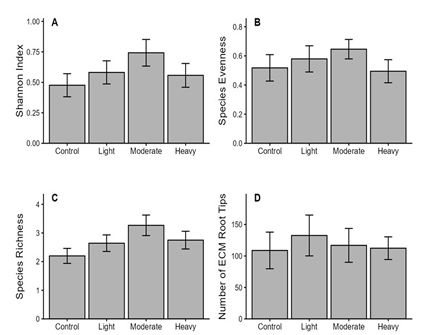
Fig. 2. Effects of thinning intensity on ectomycorrhizal (ECM) fungal community. A: Shannon diversity index, B: Evenness index, C: Species richness of ECM morphotypes, and D: Number of ECM root tips across four thinning intensities. Bars represent means ± standard errors of three plots per treatment.
One-way ANOVA (Fig. 2) indicated no statistically significant differences between the control and thinned plots in terms of alpha-diversity metrics. Shannon diversity was the lowest under the control and increased with thinning, with the highest diversity under moderate thinning. Evenness showed a similar pattern, peaking under moderate thinning, and species richness followed the same trend. Although these differences were not statistically significant, consistent directional trends suggest that moderate thinning may enhance ECM fungal diversity. In contrast, the abundance of ECM root tips was the highest under light thinning, implying that low-level canopy opening may increase the abundance of colonized tips without necessarily maximizing taxonomic diversity. Overall, compared with the control, thinning tended to increase species diversity, richness, and colonization metrics, with the strongest trends generally observed under moderate-intensity thinning. These results are consistent with the intermediate disturbance hypothesis, which proposes that moderate disturbance reduces belowground competition and increases environmental heterogeneity, thereby promoting higher diversity of ECM fungi [17,18]. From a forest management perspective, this suggests the need for a balanced approach between “non-intervention conservation” for protected areas and “intensive disturbance” for timber production. In particular, moderate thinning may serve as an effective strategy for maintaining forest health while supporting ECM diversity.
When thinning methods were compared under moderate-intensity thinning (Fig. 3), selection thinning resulted in the highest species diversity and evenness, whereas mechanical thinning produced the greatest number of ECM root tips. These results indicate that selection thinning favors a more balanced ECM fungal community, whereas mechanical thinning promotes root-tip proliferation, likely reflecting differences in soil disturbance and resource availability. Both thinning methods differed significantly from the control in several metrics (e.g., richness and tip abundance), suggesting that stand structural changes associated with thinning can modulate ECM assemblages beyond the effect of intensity alone. Because the thinning methods were sampled at a single intensity, these contrasts should be interpreted with caution.
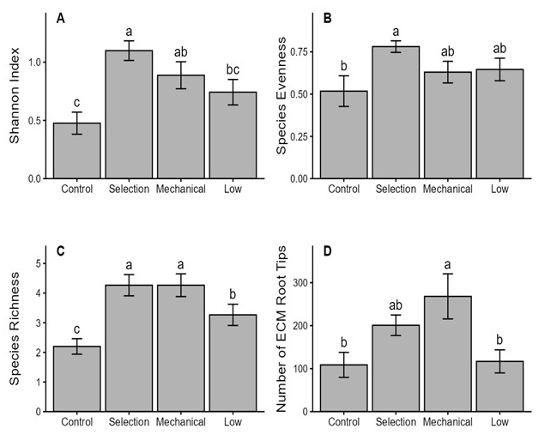
Fig. 3. Effects of thinning method on ectomycorrhizal (ECM) fungal community. A: Shannon diversity index; B: Evenness index; C: Species richness of ECM morphotypes; D: Number of ECM root tips across four thinning intensities. Bars represent means ± standard errors of three plots per treatment. Different letters above the bars indicate statistically significant differences among thinning methods (Fisher’s least significant difference test, P < 0.05).
Ecologically, thinning influences not only tree growth but also belowground fungal diversity, which is integral to long-term forest stability and ecosystem functioning [9,19]. From a management perspective, our results suggest that moderate thinning intensity and, where suitable, selection thinning, sustain forest health while supporting ECM diversity. Because the thinning methods were evaluated at only one intensity, the experimental design was unbalanced with respect to the operation type. Future studies should incorporate factorial designs spanning multiple thinning intensities and methods as well as temporal resampling to assess the persistence and generality of these patterns.
The combined evidence indicates that thinning alters ECM fungal community structure by changing both the host environment (stand density and canopy conditions) and abiotic conditions (light, soil moisture, and soil nutrients), which in turn influence enzyme expression, nutrient-uptake strategies, and interactions with hosts and other soil microbes. The outcomes of thinning vary across treatments [20] and do not act uniformly on all species within ECM fungal communities [21]; species-specific responses lead to shifts in relative abundance and changes in dominant taxa [22,23]. Taken together, varying thinning intensities and methods differentially affect the ecology of particular ECM species, thereby restructuring the community composition. We anticipate that this study will serve as a baseline for planned thinning programs in Korea and support strategies for establishing healthy forest stands.
No conflict of interest was reported by the authors.
This study was supported by a grant from the R&D Program for Forest Science Technology of the Korea Forest Service, and by a grant from the National Institute of Ecology (Project No. NIE-A-2025-01), which is funded by the Ministry of Environment of the Republic of Korea.
1. Allen MF. The ecology of mycorrhizae. Cambridge: Cambridge University Press; 1991.
2. Buscot F, Munch JC, Charcosset JY, Gardes M, Nehls U, Hampp R. Recent advances in exploring physiology and biodiversity of ectomycorrhizas highlight the functioning of these symbioses in ecosystems. FEMS Microbiol Rev 2000;24:601-14. https://doi.org/10.1111/j.1574-6976.2000.tb00561.x
[DOI]
3. Anderson IC, Cairney JWG. Ectomycorrhizal fungi: Exploring the mycelial frontier. FEMS Microbiol Rev 2007;31:388-406. https://doi.org/10.1111/j.1574-6976.2007.00073.x
[DOI]
4. Smith SE, Read DJ. Mycorrhizal symbiosis. 3rd ed. London: Academic Press; 2008.
5. Browning MHR, Whitney RD. Field performance of black spruce and jack pine inoculated with selected species of ectomycorrhizal fungi. Can J For Res 1992;22:1974-82. https://doi.org/10.1139/x92-257
[DOI]
6. Lee D, Choi J. Stand structure and sapling growth characteristics of Korean red pine stands regenerated by the seed tree method. J Korean Soc For Sci 2021;110:678-88. https://doi.org/10.14578/jkfs.2021.110.4.678
[DOI]
7. Park H, Han H, Kang KS, Hong S. Changes and prospects for pine-mushroom productivity in the Republic of Korea. J Korean Soc For Sci 2024;113:282-91. https://doi.org/10.14578/jkfs.2024.113.3.282
[DOI]
8. Lee KJ, Kim YS. A comparative study on the composition of ectomycorrhizal fungi in pine and poplar stands. Kor J Mycol 1983;11:9-13.
9. Choi JW, Lee EH, Eo JK, Koo CD, Eom AH. Community changes of ectomycorrhizal fungi by thinning in a forest of Korea. Kor J Mycol 2014;42:133-7. https://doi.org/10.4489/KJM.2014.42.2.133
[DOI]
10. Ruiz-Peinado R, Bravo-Oviedo A, López-Senespleda E, Montero G, Río M. Do thinnings influence biomass and soil carbon stocks in Mediterranean maritime pinewoods? Eur J Forest Res 2013;132:253-62. https://doi.org/10.1007/s10342-012-0672-z
[DOI]
11. Ares A, Neill AR, Puettmann KJ. Understory abundance, species diversity and functional attribute response to thinning in coniferous stands. For Ecol Manag 2010;260:1104-13. https://doi.org/10.1016/j.foreco.2010.06.023
[DOI]
12. Agerer R. Colour atlas of ectomycorrhizae. Schwäbisch Gmünd: Einhorn-Verlag Eduard Dietenberger GmbH; 1997.
13. Taylor DL, McCormick MK. Internal transcribed spacer primers and sequences for improved characterization of basidiomycetous orchid mycorrhizas. New Phytol 2008;177:1020-33. https://doi.org/10.1111/j.1469-8137.2007.02320.x
[DOI]
14. Kumar S, Stecher G, Tamura K. MEGA7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 2016;33:1870-4. https://doi.org/10.1093/molbev/msw054
[DOI]
15. Magurran AE. Ecological diversity and its measurement. Dordrecht: Springer; 1988. p. 1-5. https://doi.org/10.1007/978-94-015-7358-0
[DOI]
16. Pielou EC. An introduction to mathematical ecology. New York: John Wiley & Sons Inc; 1969.
17. Connell JH. Diversity in tropical rain forests and coral reefs: high diversity of trees and corals is maintained only in a nonequilibrium state. Science 1978;199:1302-10. https://doi.org/10.1126/science.199.4335.1302
[DOI]
18. Huston M. A general hypothesis of species diversity. Am Nat 1979;113:81-101. https://doi.org/10.1086/283366
[DOI]
19. Park YW, Kim JG, Lee H. Effect of thinning ratio on the forest environment and fruiting of ectomycorrhizal mushrooms in a Pinus densiflora stand. J Mushrooms 2023;21:22-32. https://doi.org/10.14480/JM.2023.21.1.22
[DOI]
20. Teste FP, Lieffers VJ, Strelkov SE. Ectomycorrhizal community responses to intensive forest management: Thinning alters impacts of fertilization. Plant Soil 2012;360:333-47. https://doi.org/10.1007/s11104-012-1231-6
[DOI]
21. Shaw PJ, Kibby G, Mayes J. Effects of thinning treatment on an ectomycorrhizal succession under Scots pine. Mycol Res 2003;107:317-28. https://doi.org/10.1017/S0953756203007238
[DOI]
22. Buée M, Vairelles D, Garbaye J. Year-round monitoring of diversity and potential metabolic activity of the ectomycorrhizal community in a beech (Fagus silvatica) forest subjected to two thinning regimes. Mycorrhiza 2005;15:235-45. https://doi.org/10.1007/s00572-004-0313-6
[DOI]
23. Mosca E, Montecchio L, Scattolin L, Garbaye J. Enzymatic activities of three ectomycorrhizal types of Quercus robur L. in relation to tree decline and thinning. Soil Biol Biochem 2007;39:2897-904. https://doi.org/10.1016/j.soilbio.2007.05.033
[DOI]
Table S1. Relative abundances (%) of ectomycorrhizal fungal taxa across thinning treatments
table