1. Ihm BS, Lee JS, Kim MH, Kim HS. A comparative study on the habitat of Abies koreana Wilson between Mt. Jiri and Mt. Halla. Plant Res 2000;3:138–47.
2. Jeong M, Tagele SB, Kim MJ, Ko SH, Kim KS, Koh JG, Jung DR, Jo Y, Jung Y, Park YJ, et al. The death of Korean fir (Abies koreana) affects soil symbiotic fungal microbiome: Preliminary findings. Front For Glob Change 2023;5:1114390. https://doi.org/10.3389/ffgc.2022.1114390
3. Kim YS, Chang CS, Kim CS, Gardner M. Korea fir (Abies koreana). The IUCN Red List of Threatened Species 2011: e.T31244A9618913 [Internet]. Gland: International Union for Conservation of Nature and Natural Resources; 2011 [cited 2025 Dec 18]. Available from https://dx.doi.org/10.2305/IUCN.UK.2011-2.RLTS.T31244A9618913.en
4. Koo KA, Kim DB. Review forty-year studies of Korean fir (Abies koreana Wilson). Korean J Environ Ecol 2020;34:358–71. https://doi.org/10.13047/KJEE.2020.34.5.358
5. Koo KA, Park WK, Kong WS. Dendrochronological analysis of Abies koreana W. at Mt. Halla, Korea: Effects of climate change on the growths. Korean J Ecol 2001;24:281–8.
6. Lim JH, Woo SY, Kwon MJ, Chun JH, Shin JH. Photosynthetic capacity and water use efficiency under different temperature regimes on healthy and declining Korean fir in Mt. Halla. J Korean Soc For Sci 2006;95:705–10.
7. Bais HP, Weir TL, Perry LG, Gilroy S, Vivanco JM. The role of root exudates in rhizosphere interactions with plants and other organisms. Annu Rev Plant Biol 2006;57:233–66. https://doi.org/10.1146/annurev.arplant.57.032905.105159
8. Rodriguez RJ, Redman RS, Henson JM. The role of fungal symbioses in the adaptation of plants to high stress environments. Mitig Adapt Strateg Glob Change 2004;9:261–72. https://doi.org/10.1023/B:MITI.0000029922.31110.97
9. Jeon SM, Ka KH. Cultural characteristics of Korean ectomycorrhizal fungi. Kor J Mycol 2015;43:1–12. https://doi.org/10.4489/KJM.2015.43.1.1
10. You YH, Park JM, Ku YB, Jeong TY, Lim K, Shin JH, Kim JS, Hong JW. Fungal microbiome of alive and dead Korean fir in its native habitats. Mycobiology 2024;52:68–84. https://doi.org/10.1080/12298093.2024.2307117
11. Kim CS, Jo JW, Lee H, Kwag YN, Cho SE, Oh SH. Comparison of soil higher fungal communities between dead and living Abies koreana in Mt. Halla, the Republic of Korea. Mycobiology 2020;48:364–72. https://doi.org/10.1080/12298093.2020.1811193
12. Ko YM, Gang GH, Jung DH, Kwak YS. Comparative analysis of rhizosphere fungal communities in Korean fir trees. Mycobiology 2024;52:287–97. https://doi.org/10.1080/12298093.2024.2397857
13. Sang Y, Jin L, Zhu R, Yu XY, Hu S, Wang BT, Ruan HH, Jin FJ, Lee HG. Phosphorus-solubilizing capacity of Mortierella species isolated from rhizosphere soil of a poplar plantation. Microorganisms 2022;10:2361. https://doi.org/10.3390/microorganisms10122361
14. Ozimek E, Hanaka A. Mortierella species as the plant growth-promoting fungi present in the agricultural soils. Agriculture 2021;11:7. https://doi.org/10.3390/agriculture11010007
15. Li F, Zhang S, Wang Y, Li Y, Li P, Chen L, Jie X, Hu D, Feng B, Yue K, et al. Rare fungus, Mortierella capitata, promotes crop growth by stimulating primary metabolisms related genes and reshaping rhizosphere bacterial community. Soil Biol Biochem 2020;151:108017. https://doi.org/10.1016/j.soilbio.2020.108017
16. Osorio Vega NW, Habte M, León Peláez JD. Effectiveness of a rock phosphate solubilizing fungus to increase soil solution phosphate impaired by the soil phosphate sorption capacity. Rev Fac Nac Agron Medellín 2015;68:7627–36. https://doi.org/10.15446/rfnam.v68n2.50950
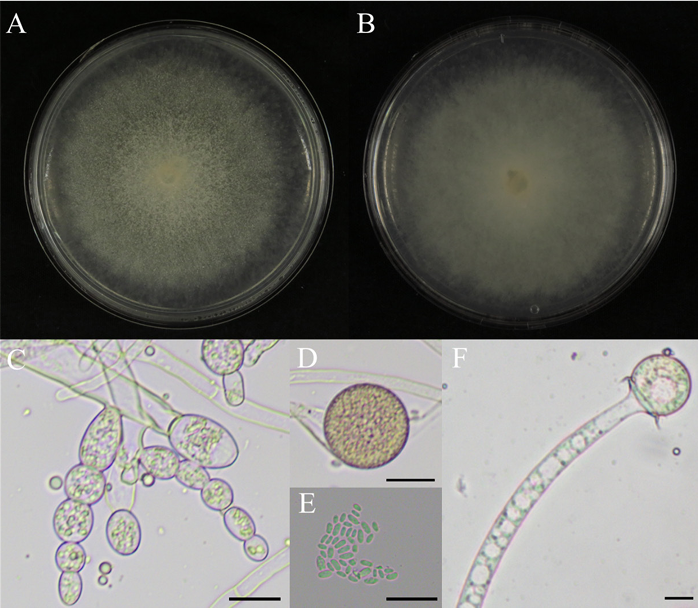
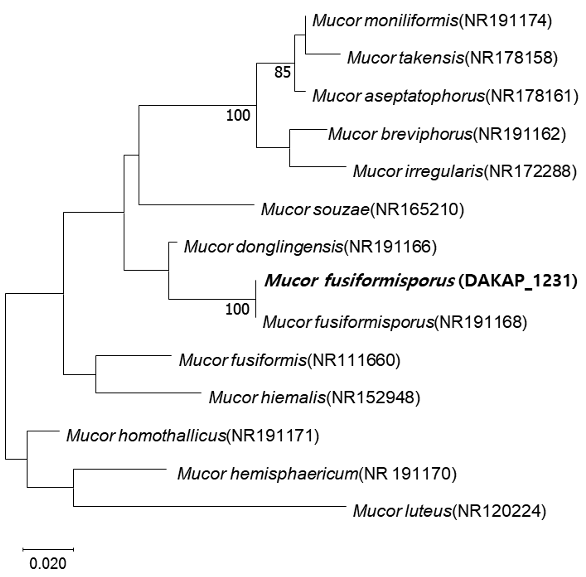
17. Nguyen TTT, Jeon YJ, Mun HY, Goh J, Chung N, Lee HB. Isolation and characterization of four unrecorded Mucor species in Korea. Mycobiology 2020;48:29-36. https://doi.org/10.1080/12298093.2019.1703373
18. Ding ZY, Ji XY, Li F, Liu WX, Wang S, Zhao H, Liu XY. Unveiling species diversity within early-diverging fungi from China IX: Four new species of Mucor (Mucoromycota). J Fungi 2025;11:682. https://doi.org/10.3390/jof11090682
19. Schipper MAA. On Mucor mucedo, Mucor flavus and related species. Stud Mycol 1975;10:1–33.
20. Hussein KA, Joo JH. Zinc ions affect siderophore production by fungi isolated from the Panax ginseng rhizosphere. J Microbiol Biotechnol 2019;29:105–13. https://doi.org/10.4014/jmb.1712.12026
21. Kuhnert R, Oberkofler I, Peintner U. Fungal growth and biomass development is boosted by plants in snow-covered soil. Microb Ecol 2012;64:79–90. https://doi.org/10.1007/s00248-011-0001-y
22. Telagathoti A, Probst M, Peintner U. Habitat, snow-cover and soil pH affect the distribution and diversity of Mortierellaceae species and their associations to bacteria. Front Microbiol 2021;12:669784. https://doi.org/10.3389/fmicb.2021.669784
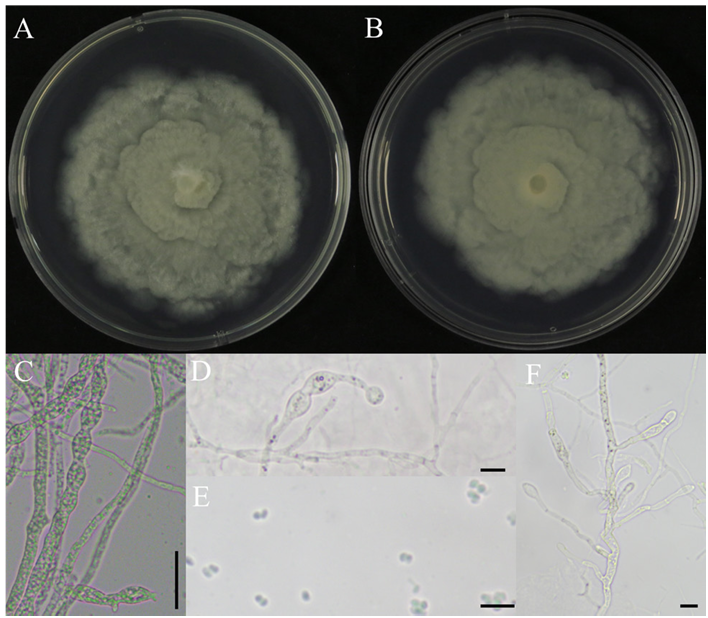
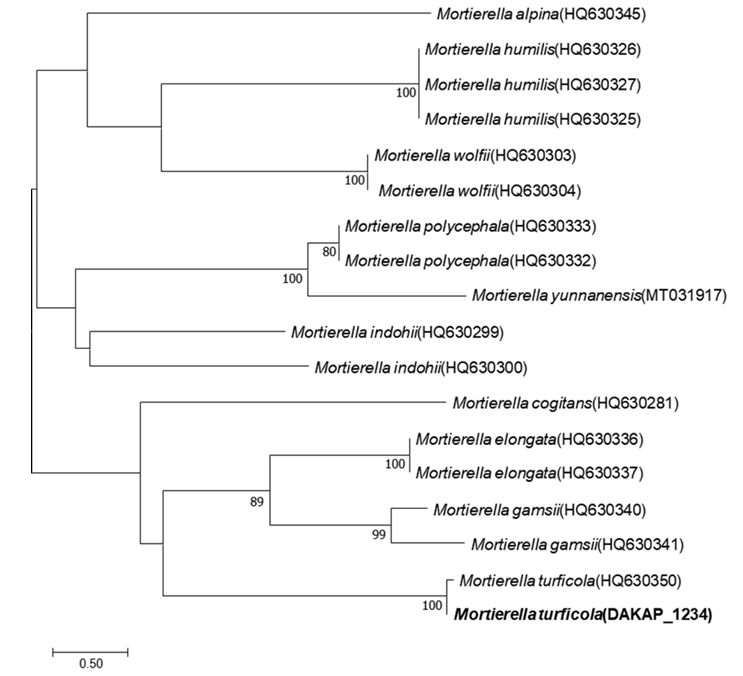
23. Telagathoti A, Probst M, Mandolini E, Peintner U. Mortierellaceae from subalpine and alpine habitats: New species of Entomortierella, Linnemannia, Mortierella, Podila and Tyroliella gen. nov. Stud Mycol 2022;103:25–58. https://doi.org/10.3114/sim.2022.103.02
24. Wagner L, Stielow B, Hoffmann K, Petkovits T, Papp T, Vágvölgyi C, de Hoog GS, Verkley G, Voigt K. A comprehensive molecular phylogeny of the Mortierellales (Mortierellomycotina) based on nuclear ribosomal DNA. Persoonia 2013;30:77–93. https://doi.org/10.3767/003158513×666268
25. White TJ, Bruns TD, Lee SB, Taylor JW. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ, editors. PCR protocols: A guide to methods and applications. San Diego: Academic Press; 1990. p. 315–22. https://doi.org/10.1016/B978-0-12-372180-8.50042-1
26. Gardes M, Bruns TD. ITS primers with enhanced specificity for basidiomycetes – application to the identification of mycorrhizae and rusts. Mol Ecol 1993;2:113–8. https://doi.org/10.1111/j.1365-294x.1993.tb00005.x
27. O’Donnell K. Fusarium and its near relatives. In: Reynolds DR, Taylor JW, editors. The fungal holomorph: Mitotic, meiotic and pleomorphic speciation in fungal systematics. Wallingford: CAB International; 1993. p. 225–33.
28. Kumar S, Stecher G, Li M, Knyaz C, Tamura K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol 2018;35:1547–9. https://doi.org/10.1093/molbev/msy096
29. Takashima Y, Degawa Y, Ohta H, Narisawa K. Mortierella sugadairana, a new homothallic species related to the firstly described heterothallic species in the genus. Mycoscience 2018;59:200–5. https://doi.org/10.1016/j.myc.2017.10.004