Wasabi (Eutrema japonicum (Miq.) Koidz.) is a perennial plant belonging to the family Brassicaceae, and its native range includes Japan, Korea, and Sakhalin [1]. This plant grows primarily in temperate regions and is used worldwide as a salad or cooking sauce. In Korea, this plant is cultivated as a vegetable in vinyl greenhouses in the alpine areas.
In 2019 and 2021, we observed gray mold symptoms on leaves of wasabi plants grown in vinyl greenhouses in Taebaek and Pyeongchang, Gangwon Province, Korea, during crop disease surveys. The symptoms appeared at the edges of the leaves, where the infected areas turned black and rotted, and many gray molds formed on the lesions (Fig. 1A and B). One hundred plant leaves from each vinyl greenhouse were investigated for the incidence of gray mold. The disease incidence on leaves in the vinyl greenhouses at the two locations ranged from 1 to 30% (Table 1).
Wasabi leaves with gray mold symptoms were collected from vinyl greenhouses at two locations, and fungi were isolated from the diseased leaves. A conidial suspension was prepared from conidial masses on a gray mold using sterile distilled water and streaked on 2% water agar (WA) plates using a sterile loop. After incubation of the WA plates for 24 hr
Fig. 1
Gray mold symptoms of wasabi plants. (A and B) Symptoms observed on the leaves in the investigated vinyl greenhouse. (C) Symptoms on the leaves induced by artificial inoculation test with Botrytis cinerea isolate (EJBC-2102). (D) A non-inoculated plant (control).

Table 1
Occurrence of gray mold on wasabi leaves in vinyl greenhouses located in Taebaek and Pyeongchang, Korea in 2019 and 2021

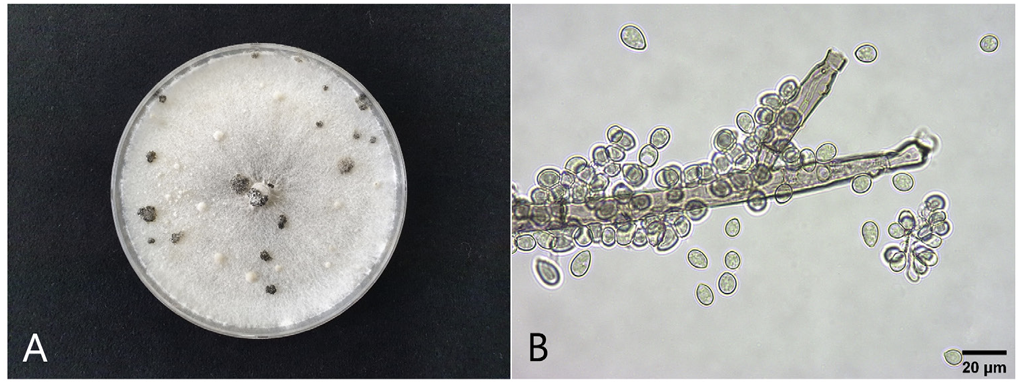
The four fungal isolates (EJBC-1901, EJBC-1902, EJBC-2101, and EJBC-2102) were cultured on PDA in 9 cm diameter Petri dishes at 20°C in the dark for 25 days to investigate their cultural and morphological characteristics. The colonies of the isolates cultured on PDA consisted of white to gray mycelia and black, spherical or irregular sclerotia with diameters of 1‒7mm (Fig. 2A). Fifty conidia and 25 conidiophores from each isolate were examined for morphology. Conidia were hyaline to pale brown, globose or ellipsoidal, and measured 5.5‒13.2 × 5.0‒10.2 μm (av. 10.0 × 7.3 μm) (Fig. 2B). Conidiophores were erect, more than 1mm in length, 11‒20 μm thick, branched, brown below, and paler near the apex. The morphological characteristics of the isolates were similar to those of Botrytis cinerea Pers. as described previously [2].
Fig. 2
Cultural and morphological features of Botrytis cinerea isolates from diseased wasabi leaves. (A) A colony of an isolate (EJBC-2102) grown on potato dextrose agar at 20°C for 25 days. (B) A conidiophore and conidia. .
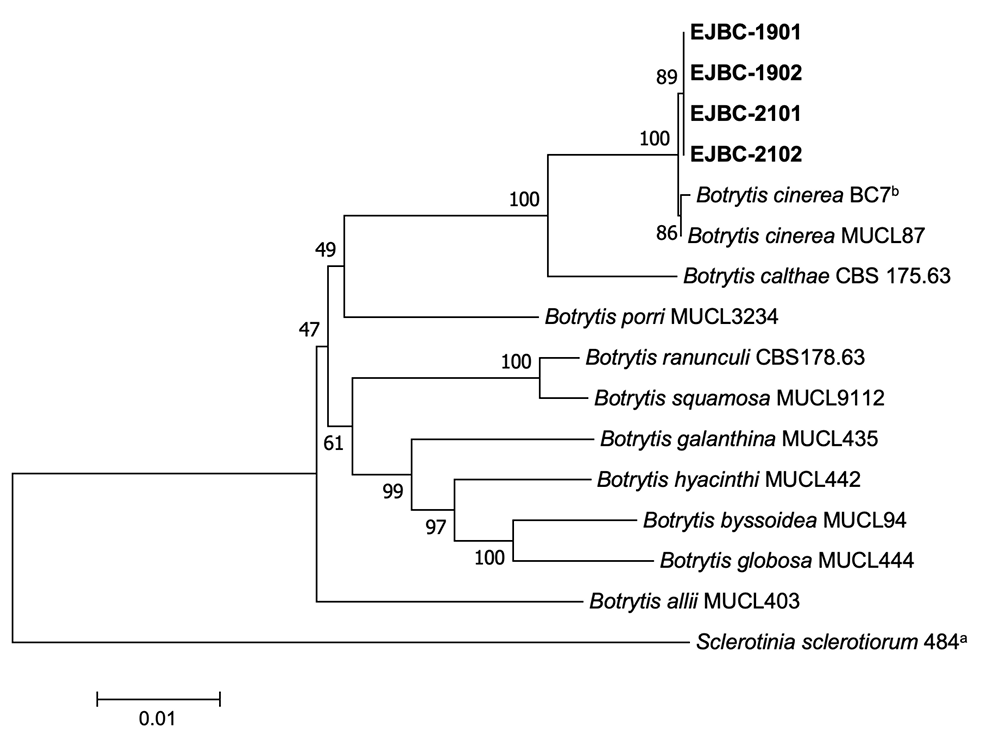
Phylogenetic analysis was conducted to confirm the identity of B. cinerea isolates based on their morphological characteristics. Genomic DNA of the isolates was extracted using a previously described protocol [3], with slight modifications. Polymerase chain reaction (PCR) products of the glyceraldehyde-3-phosphate dehydrogenase (G3PDH), heat-shock protein 60 (HSP60), and DNA-dependent RNA polymerase subunit II (RPB2) gene regions were obtained using primer sets with amplification conditions described in a previous study [4] and DNA Free-Multiplex Master Mix (Cellsafe, Korea). PCR products were purified using a Universal DNA Purification Kit (Tiangen, Beijing, China). PCR products were sequenced at Bionics Co., Ltd. (Seoul, Korea) with the same primers. Sequences were adjusted using SeqMan II (DNASTAR Inc., Madison, WI, USA) if necessary. Sequence alignment of the isolates from wasabi and other Botrytis spp. [4] was performed using MUSCLE [5]. Sclerotinia sclerotiorum 484 [6] was selected as the outgroup taxon. MEGA version 7 software [7] was used to process and enhance multiple sequence alignments. Neighbor-joining analysis for concatenated alignments was conducted with maximum composite likelihood model performing 1,000 bootstrap replicates using MEGA version 7 software [7]. Phylogenetic analysis revealed that all the isolates clustered with two strains (BC7b and MUCL87) of B. cinerea (Fig. 3). The sequence data of G3PDH, HSP60, and RPB2 obtained from the four isolates were deposited in GenBank under the accession numbers OR469314‒OR469317, OR469322‒OR469325, and OR469318‒OR469321, respectively.
Fig. 3
Phylogenetic tree based on glyceraldehyde-3-phosphate dehydrogenase, heat-shock protein 60, and DNA-dependent RNA polymerase subunit II gene regions of the four isolates (EJBC-1901, EJBC-1902, EJBC2101, and EJBC-2102) from wasabi and reference strains of Botrytis species. Sequences of the reference species were obtained from the NCBI GenBank database. The phylogenetic tree was generated using the neighborjoining method with a maximum composite likelihood model. The bootstrap support values are shown at the nodes. The scale bar represents the number of nucleotide substitutions per site.
Two isolates of B. cinerea (EJBC-1902 and EJBC-2102) were tested for pathogenicity on wasabi leaves using artificial inoculation. A conidial suspension (1‒2×106 conidia/ mL) of each isolate was prepared from the 30-day-old PDA cultures. Then, 30 mL of the conidial suspension of each isolate was sprayed onto the leaves of a 10-month-old wasabi plant grown in a circular plastic pot (height: 15 cm; upper diameter: 17 cm; lower diameter: 10 cm) in a vinyl greenhouse. A control wasabi plant was treated with 30 mL of sterile distilled water. The inoculated plants were placed in plastic boxes (72×54×45 cm) under 100% relative humidity in a cultivation room at 18‒22°C for 3 days. The inoculated plants were removed from the boxes and placed in the cultivation room. The pathogenicity of the isolates was rated based on the degree of gray mold symptoms 7 days after inoculation. The inoculation tests were conducted in triplicates. All tested isolates caused gray mold symptoms in the leaves of inoculated plants (Fig. 1C), but no symptoms were observed in the leaves of control plants (Fig. 1D). The symptoms were similar to those observed in the wasabi leaves in the investigated vinyl greenhouses. Re-isolation of the inoculated isolates from the lesions was confirmed.
B. cinerea causes gray mold in various plants, including many of economic importance, such as bean, lettuce, gooseberry, tomato, and vine [2]. This fungus has been reported to cause gray mold or Botrytis leaf blight in wasabi in other countries [8,9]. The occurrence of gray mold caused B. cinerea has been reported in many plants in Korea [10]. However, there have been no reports on occurrence of gray mold in wasabi in Korea. This is the first report of B. cinerea causing gray mold in wasabi in Korea.


